 The bond angle is 90° in one plane and 120° in another plane. The central atom is surrounded by five atoms – three in one plane and two on opposite sides of the plane. Unlike the trigonal planar, the trigonal bipyramidal structure is three-dimensional. Trigonal Bipyramidal: The molecule’s shape resembles a pyramid with a triangular base. This shape is because the four bond pairs experience minimum repulsion when the bonds are directed toward the corners of the tetrahedron. Tetrahedral: This shape occurs when one atom occupies the center, and four others are located at the corners of a tetrahedron. Examples are boron trifluoride (BF 3), boron trichloride (BCl 3), and sulfur trioxide (SO 3).ģ. It has one atom at the center and three at the corners of an equilateral triangle, making a bond angle of 120°. Trigonal Planar: The molecule forms a triangular shape in one plane. Examples of molecules with linear geometry are carbon dioxide (CO 2), beryllium chloride (BeCl 2), and nitric oxide (NO).Ģ. The atoms are arranged in a straight line, and the angle between the bonds is 180 °. Linear: It refers to the geometry shaped by a central atom surrounded by two other atoms. 
The VSEPR theory describes five fundamental shapes of molecules. The molecule’s energy will be at its lowest, thereby increasing stability. Since the electron pairs repel each other, they will remain at a maximum distance.For resonance structures, the VSEPR theory is individually applied to each structure. 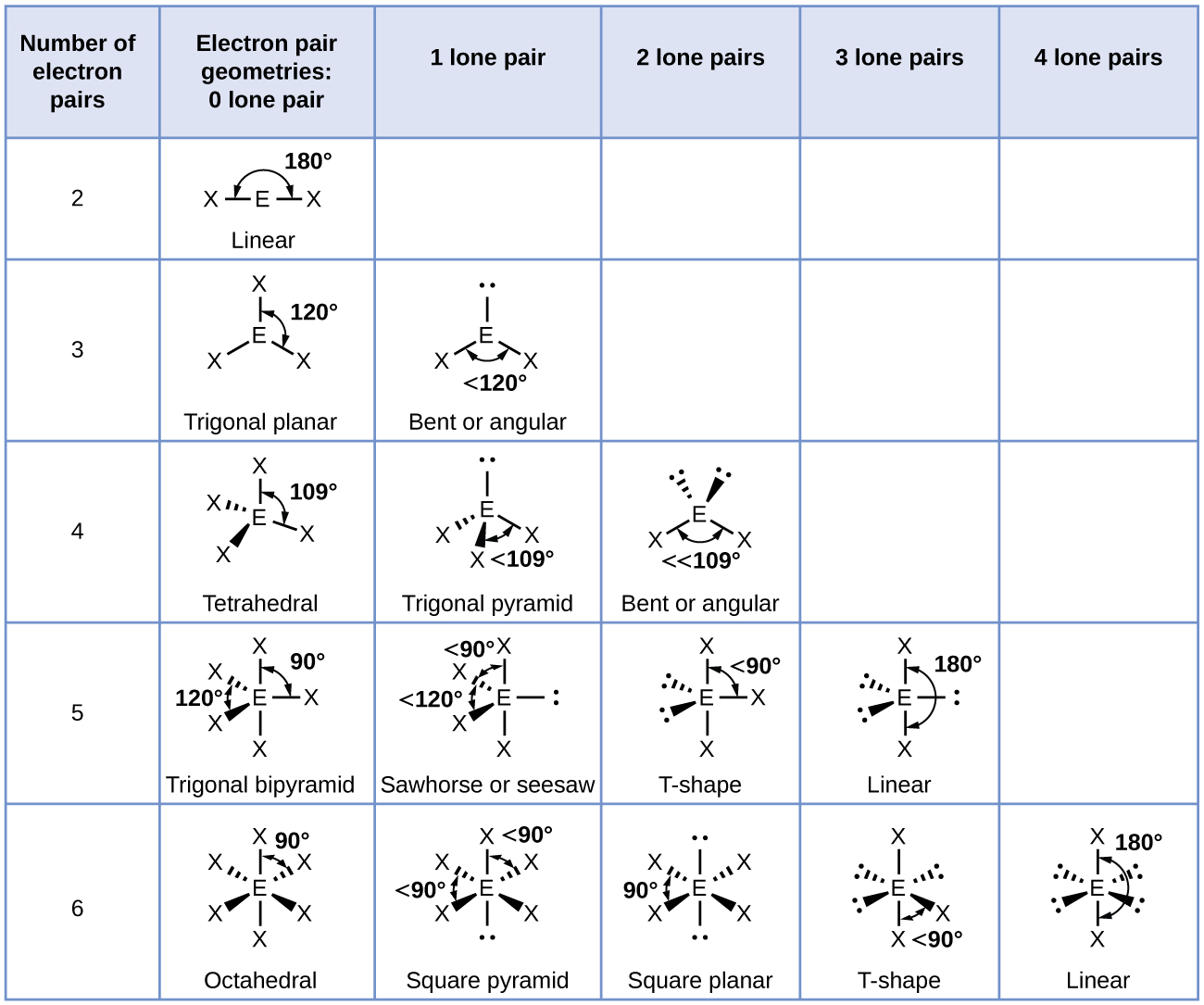
As a result, the shape is distorted, and the bond angles are reduced. When lone pairs are introduced into the basic structure, they squeeze the bond pairs closer together.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
